U.S. Government: DNA Collected from Newborn Dried Blood Spots No Longer Protected From Being Used in Human Research
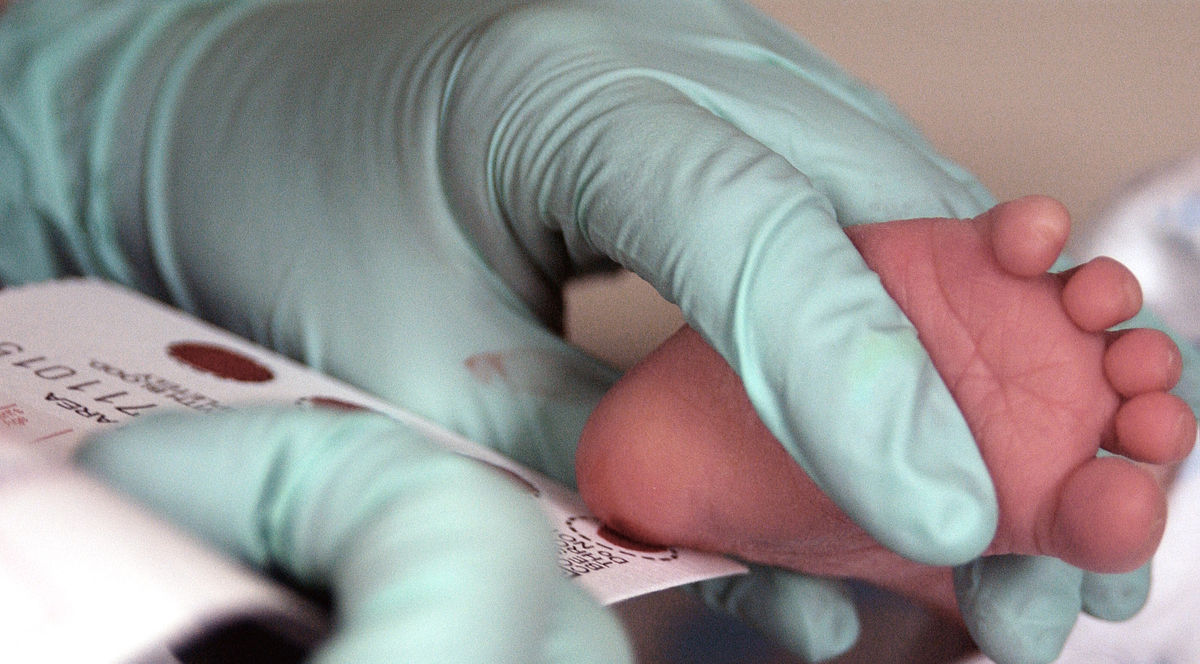
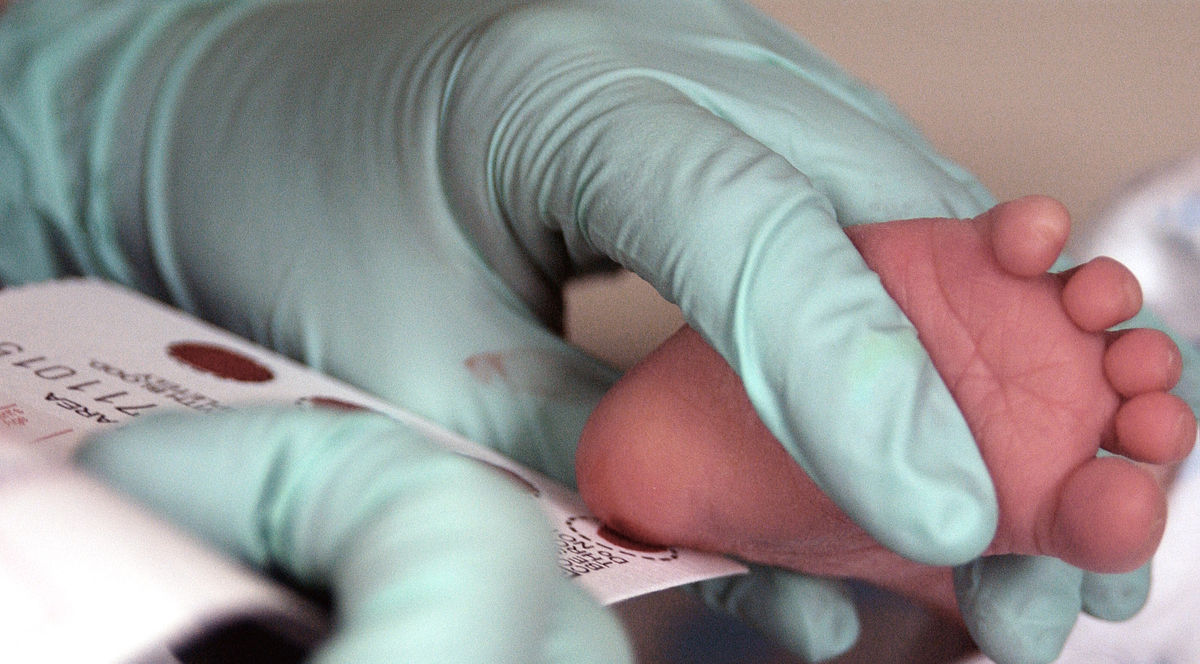
The blood of a two week-old infant is collected for a Phenylketonuria, or PKU, screening. Image Source.
Baby DNA Will No Longer be Protected by Previously Enacted Consent Requirements
by Citizens’ Council for Health Freedom
READ MORE AT HEALTH IMPACT NEWS
ST. PAUL, Minn.—The Office for Human Research Protections at the U.S. Department of Health and Human Services used a Frequently Asked Questions (FAQ) to issue a notice on federally funded research using newborn dried blood spots taken by states for genetic testing at birth.
Citizens’ Council for Health Freedom (CCHF) first discovered long-term state warehousing of newborn DNA in 2003 and has been educating Americans on the fact that some states store and use newborn blood spots without parental consent. CCHF has actively engaged parents to protect their newborn baby’s DNA and genetic privacy rights.
CCHF president and co-founder Twila Brase released the following statement in response to the announcement:
Today, the U.S. Department of Health and Human Services gave notice that newborn DNA will no longer be protected by parent consent requirements enacted in 2014.
In an FAQ document, HHS says the 2014 federal law’s consent requirements for federally funded research and its requirement to treat research using the newborn’s DNA as ‘human subjects research’ no longer apply and have not applied since July 19, 2018, the effective date of the revised federal ‘Common Rule’ on federally funded research.
Newborns cannot protect themselves from genetic exploitation.
Congress was right in 2014 to protect these newborn citizens from the taxpayer-funded genetic analysis, research and profiteering that were taking place, and will now take place again, without the consent of their parents.
These dried blood spots, taken at birth as part of the state newborn genetic testing program in all 50 states, and stored indefinitely in some states, are prized for the DNA they hold. Texas has sold them and used them for barter. California sells them for about $20 to $40 per spot.
Parents in Indiana, Michigan, Minnesota, and Texas have filed lawsuits, forcing several state health departments to destroy the newborn DNA blood spot cards they had stored for decades, and sometimes shared and used, without parent consent. The parent lawsuit in Minnesota went all the way to the state supreme court before the parents won.
Congress should act now to restore the parent consent requirements that were in place for more than three years and the HHS Office for Human Research Protections have dismantled. The genetic privacy rights of the four million babies born in America every year are at stake.
The Office for Human Research Protections (OHRP) posted this question and answer as an FAQ:
Section 12 of the Newborn Screening Saves Lives Reauthorization Act of 2014 changed the ability of investigators to conduct certain federally funded research with newborn dried blood spots regardless of the identifiability of the blood spots. Are these requirements of the Act still in effect?
No. Research with nonidentified newborn dried blood spots, similar to other research with nonidentified biospecimens, is not considered research with human subjects under both the 2018 and pre-2018 Requirements, and thus, is not subject to 45 CFR part 46.
Although Section 12 of the Newborn Screening Saves Lives Reauthorization Act (NSSLRA) of 2014 stated that certain federally funded research on newborn dried blood spots would be considered human subjects research (regardless of the identifiability of the blood spots), and prohibited IRBs from waiving consent for such research, Section 12 applied only until changes to the Common Rule were promulgated.
Thus, as of the effective date of the revised Common Rule (July 19, 2018), the requirements in Section 12 of the NSSLRA of 2014 are no longer in effect.
Brase writes extensively about protecting Baby DNA, as well as socialized medicine, patient privacy, electronic health records, health freedom and how the Affordable Care Act has harmed patients and doctors since 2010 in the new book “Big Brother in the Exam Room: The Dangerous Truth About Electronic Health Records.” For more information, visit BigBrotherintheExamRoom.com.
Learn more about CCHF at www.cchfreedom.org








While I sill remember …
Yesterday I was listening to the reading for the blind radio station …
Apparently — the Australian government are going to have new assessments or children with autism & ADHD
WHY ??
Because the numbers are too high.
There will also be a reworking of the monies allocated to the care of these afflicted children …. DOWNWARDS OF COURSE.
The actual article is in one of mainstream local papers – the Heral Sun – The Australian etc health section.