Challenging Influenza

“Our review found no human experimental studies published in the English-language literature delineating person-to-person transmission of influenza.“
Bridges et al. 2003 https://academic.oup.com/cid/article/37/8/1094/2013282?login=false
Last year, I took a look at the first ever human challenge trial for “SARS-COV-2” and showed why the results are fraudulent and completely pseudoscientific. Within the article, I provided an explanation from the WHO as to what a human challenge trail is and why these studies are considered mostly unethical today.
“Infectious human challenge studies involve deliberate exposure of human volunteers to infectious agents. Human challenge studies have been conducted over hundreds of years and have contributed vital scientific knowledge that has led to advances in the development of drugs and vaccines. Nevertheless, such research can appear to be in conflict with the guiding principle in medicine to do no harm. Well documented historical examples of human exposure studies would be considered unethical by current standards.”
Click to access Human_challenge_Trials_IK_final.pdf
Without rehashing everything here, let’s just say that the challenge studies conducted over the last 100 years, from the 1918 Rosenau Spanish flu experiments to many other similar studies, consistently failed to show human-to-human transmission of disease using the fluids of sick individuals. It was thus deemed much more ethical to create toxic soups from diseased brains and spinal cords of humans and animals and then inject these concoctions into the brain, eyes, throat, nose, stomach, veins, testicles, etc. of healthy animals. It was easier for the researchers to create experimental disease in this unnatural manner and then claim that the results seen from torturing animals were somehow relevant to humans.
As the results from human challenge studies have been consistently unsuccessful, as noted in the opening quote by Bridges et al., these studies are not performed very often. However, sometimes, we get lucky and see modern-day interpretations of these experiments using lab-manufactured cell cultured goo that is injected into the nose of willing participants. Interestingly, the most recent of these studies have both involved the same researcher, Jonathan S. Nguyen-Van-Tam. The study presented below is Mr. Nguyen-Van-Tam’s previous challenge trial that was performed prior to his “SARS-COV-2” paper that I looked at previously. In this February 2020 study that was published in June of the same year, Nguyen-Van-Tam attempted to determine and provide definitive proof for air-borne transmission of influenza A. With what can only be an amazing coincidence, this paper served as the perfect precursor to performing a “SARS-COV-2” human challenge trial a year later. I have interjected commentary throughout the study, pointing out some interesting highlights, as well as various flaws, within the paper. You will see that not only that the above statement by Bridges et al. from 2003 still remains true today, you will see why they shy away from carrying out such studies more often.
Minimal transmission in an influenza A (H3N2) human challenge-transmission model within a controlled exposure environment
To begin with, the authors immediately admit that there is still uncertainty about the importance of influenza transmission via airborne droplets. The authors openly express the fact that human-to-human transmission is poorly understood and that the importance of the varying routes of supposed transmission remains uncertain. Apparently, after over 100 years of studying the various influenza “viruses,” they still do not understand how it supposedly spreads. During this so-called challenge trial where everything is perfectly controlled, they could only show one “transmitted infection” by way of the always unreliable antibody testing. These results were not as they expected and were considered significantly lower than their proof-of-concept model, even taking into account that they had far more participants in this study and more days of “exposure.”
It is admitted by the authors that the current evidence for human-to-human transmission of influenza is based upon results aquired from “virus” deposition and survival in the environment; the epidemiology of disease; pharmaceutical and non-pharmaceutical interventions; animal models; and mathematical models of transmission. The evidence garnered from these means is said not to be conclusive. In other words, our current understanding of influenza transmission is based upon indirect evidence, and there is ZERO conclusive scientific evidence proving human-to-human transmission. Thus, it is ASSUMED that most influenza transmission occurs from symptomatic infection via large droplets at a short range. As there has been a failure to aquire definitive evidence, it was concluded that human-to-human challenge trials, which had “never been performed,” are the best way of obtaining direct definitive evidence. However, upon doing such a study, the authors admitted that this resulted in nothing but failure:
Introduction
“Uncertainty about the importance of influenza transmission by airborne droplet nuclei generates controversy for infection control. Human challenge-transmission studies have been supported as the most promising approach to fill this knowledge gap. Healthy, seronegative volunteer ‘Donors’ (n = 52) were randomly selected for intranasal challenge with influenza A/Wisconsin/67/2005 (H3N2). ‘Recipients’ randomized to Intervention (IR, n = 40) or Control (CR, n = 35) groups were exposed to Donors for four days. IRs wore face shields and hand sanitized frequently to limit large droplet and contact transmission. One transmitted infection was confirmed by serology in a CR, yielding a secondary attack rate of 2.9% among CR, 0% in IR (p = 0.47 for group difference), and 1.3% overall, significantly less than 16% (p<0.001) expected based on a proof-of-concept study secondary attack rate and considering that there were twice as many Donors and days of exposure. The main difference between these studies was mechanical building ventilation in the follow-on study, suggesting a possible role for aerosols.
Influenza virus is a pathogen of global health significance, but human-to-human transmission remains poorly understood.In particular, the relative importance of the different modes of transmission (direct and indirect contact, large droplet, and aerosols (airborne droplet nuclei)) remains uncertainduring symptomatic and asymptomatic infection [1–4].
The evidence base for influenza transmission is derived from studies that have assessed: virus deposition and survival in the environment; the epidemiology of disease; pharmaceutical and non-pharmaceutical interventions; animal models; and mathematical models of transmission. Those approaches have yet to produce conclusive data quantifying the relative importance of human-human transmission modes [1,2].
Infection control guidance for pandemic and seasonal influenza assumes that most transmission occurs during symptomatic infection, predominantly via large droplet spread at short range (1-2m) [1]. Thus, social distancing measures are often proposed to mitigate the spread and impact of a pandemic; and hand washing and respiratory etiquette are promoted to reduce transmission. Evidence to support the possibility of aerosol transmission has grown over recent years [5–7]. and leads to controversies about when and if filtering facepiece respirators (and other precautions designed to prevent inhalation of aerosols) versus surgical masks (mainly capable of reducing large droplets and some fine particles) should be used to protect healthcare workers, particularly during a severe pandemic [1,3,4,8–10].
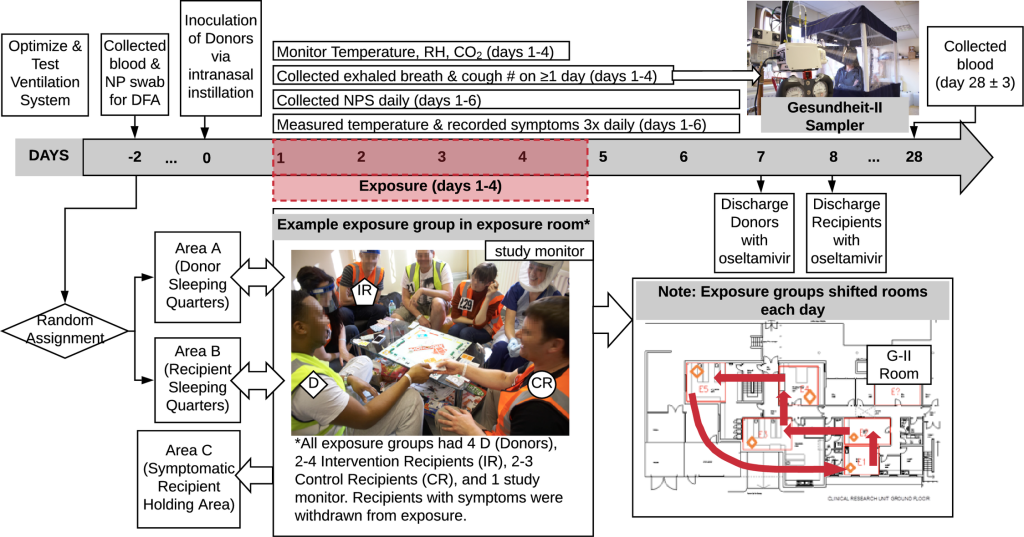
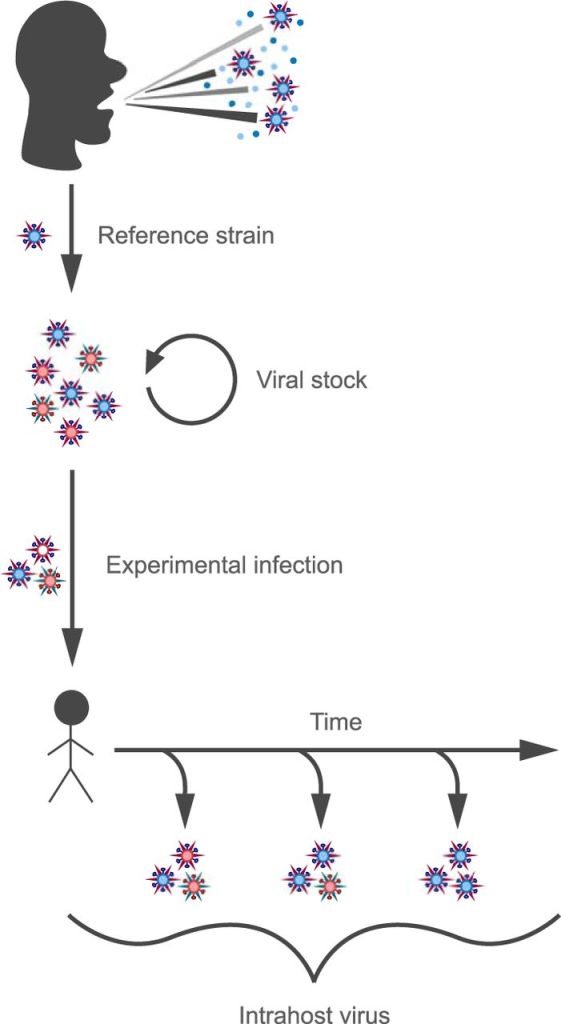
An expert panel, after in-depth review of the challenges facing community- and workplace-based intervention studies and their failure thus far to provide definitive evidence regarding the relative contribution of the various modes,concluded that a human challenge-transmission study would be a more promising direction for future research [11]. Influenza challenge studies in humans have been conducted to investigate disease pathogenesis and the efficacy of antivirals and vaccines. Challenge studies assessing human-to-human transmission had not been performed [11]. In 2009, we demonstrated proof-of-concept that healthy seronegative volunteers inoculated intranasally with influenza A/Wisconsin/67/2005 (A/WI), an H3N2 virus, would develop symptoms of influenza-like illness (ILI) and, under two days of household-like conditions without environmental controls, transmit infection to other seronegative volunteers. This suggested that larger scale human challenge-transmission models might be useful to evaluate transmission modes. A subsequent international workshop discussed the potential that human challenge-transmission studies, with appropriate interventions, monitoring of aerosol shedding, and environmental controls, could provide definitive results [12]. Here, we report a large follow-on study, including design factors (Fig 1) aimed at assessing the importance of aerosol transmission in human-to-human transmission of influenza virus. Although the study did not achieve the intended level of transmission required for a more conclusive interpretation, it has revealed important lessons about potential airborne transmission and study design that represent critical knowledge to support effective large-scale, costly studies in this area.
Results
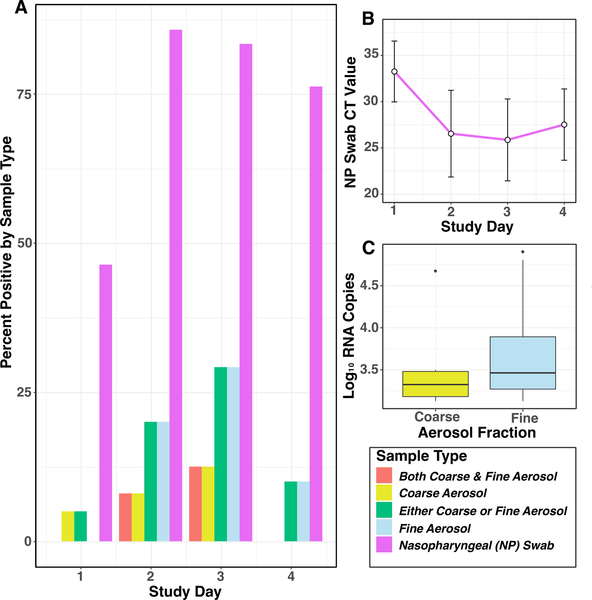
Ready for some impressive results? Let’s start off with the fact that no serious adverse events occurred in any of the participants in the study, showing that this was not a very dangerous influenza “virus.” 52 donors were subjected to intranasal inoculation of the “virus,” which is obviously not a natural route of infection. This was said to produce an “infection” in 42 of the 52 donors, which was laboratory confirmed by qRT-PCR. Of these, 25 were said to have an influenza-like illness, while 10 were classified as asymptomatic. Only 36 donors had nasopharyngeal swabs that tested positive by qRT-PCR. It was claimed that of the 42 “infected” donors, they observed aerosol shedding from just 11 of them.
Despite the “successful infections,” none of the participants developed a fever. Only a single “transmission” was noted in a control recipient, which was confirmed by way of the inaccurate antibody testing. This person was said to be symptomatic but consistently failed when tested by the “gold standard” qRT-PCR. Two other control recipients tested positive by qRT-PCR, but they did not meet the laboratory confirmation criteria. This meant that there was only one “successful transmission,” and it was not confirmed by the “gold standard.”
Participation and safety
Between January and June 2013, 496 seronegative (HAI≤10 to the challenge virus antigen) volunteers underwent study-specific screening and 166 entered the quarantine unit, of whom 127 proved suitable for final study entry (Fig 2 –trial profile; S5 Text–volunteer baseline characteristics). Thirty-nine subjects were discharged before inoculation or exposure per protocol as described in Methods. Three separate quarantine EEs (exposure events) took place in March, April, and June 2013 involving Q1: 41 (20 Donors; 11 CR; 10 IR), Q2: 31 (12 Donors; 9 CR; 10 IR) and Q3: 55 (20 Donors; 15 CR; 20 IR) subjects respectively, with 4 Donors and 4 to 7 Recipients per exposure group. No serious adverse events were recorded in volunteers who commenced the study.
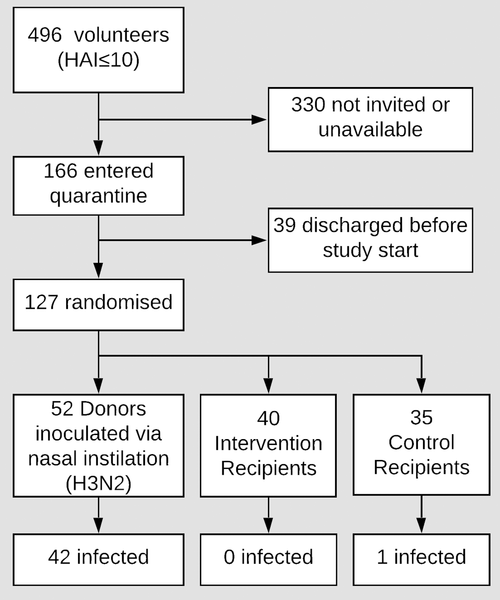
Environmental control
In Q1 relative humidity averaged 40% (Standard Deviation 9%), room temperature averaged 20.2 oC (0.4 oC) and CO2 concentration averaged 1430ppm (110ppm). For Q2 and Q3, respectively, the corresponding values were 44% (4%), 21.4°C (0.3 oC), 1810ppm (160ppm), and 57% (4%), 21.4°C (0.3 oC), 1810ppm (160ppm). Outdoor CO2 concentration proxies, taken from the average of CO2 measurements during 2:00am-3:00am were 418, 435, and 422 ppm, for Q1, Q2, and Q3, respectively.
Donor status
Donor status is summarised by quarantine study in Table 1. Over all quarantines combined, intranasal inoculation produced an infection rate of 81% (42/52) among inoculated volunteers. Of the 42 lab-confirmed infected Donors 25 (60%) had ILI and 10 (24%) were classified as asymptomatic (4 in Q1, 4 in Q2, and 2 in Q3).

Ten Donors had greater immunity on admission, as identified by samples collected on day -2 (HAI>10 or MN≥80), than at their earlier screening. Four of the 10 seroconverted (i.e. had a 4-fold rise in HAI or MN titres) between admission to quarantine and follow-up. Five of the 10 met laboratory case definition by qRT-PCR including all four who seroconverted. The one additional qRT-PCR positive Donor had positive swabs on study days 2 and 3 in Q2.
Virus shedding by donors
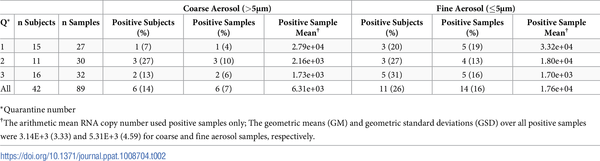
Overall, 36 Donors had nasopharyngeal swab (NPS) that tested positive by PCR for A/WI. Of these 36: 53% (n = 19) were positive on day 1 post-challenge; 94% (34) on day 2; 97% (35) on day 3; 86% (31) on day 4; 92% (33) on day 5; and 67% (24) on day 6 (Fig 3).
doi:10.1371/journal.ppat.1008704.g003
Aerosol shedding was determined for 25 Donors on day 1, 31 on day 2, 30 on day 3, and 24 on day 4, and for a total of 36 person-days in Q1, 34 person-days in Q2, and 40 person-days in Q3. Aerosol shedding from infected Donors, detected in Gesundheit-II samples, is summarised in Table 2. Six (7%) of the coarse and 14 (16%) of the fine aerosol samples had detectable viral RNA. We observed aerosol shedding from 11 (26%) of the 42 successfully infected Donors. The geometric mean (GM) and geometric standard deviation (GSD) for coarse and fine aerosol viral RNA copy numbers per 30-min sample were 3.1E+3 (3.3) and 5.3E+3 (4.6), respectively. The maximum levels of shedding into coarse and fine aerosols were 2.79E+4 and 8.02E+4 RNA copies, respectively (Fig 3).
Recipient status
Recipient status is shown in Table 3. There were similar rates of symptoms in both IR and CR groups, although more in the CR group met the study definition of ILI, the rates were not significantly different (p = 0.23); no Recipient developed fever.One infection was confirmed by serology (HAI increased from ≤10 to 40 and MN increased from 10 to 320) in a CR subject who was symptomatic and whose symptoms met the definition of ILI, but whose qRT-PCR evaluations were persistently negative. Two other CR were transiently qRT-PCR positive but neither met laboratory positivity criteria (S6 Text). Both were asymptomatic and had no change from baseline serology. Thus, there was only one, confirmed transmission event. The CR and IR group SARs (2.9% and 0%) were not significantly different (p = 0.47).
To compare these results with the SAR from the proof-of-concept study, we recomputed the latter results using the current, more stringent outcome criteria. The adjusted proof-of-concept SAR was 8.3% giving an expected SAR of 16%. The observed SAR for the current study was not significantly different than that of the adjusted SAR from the proof-of-concept study, but was significantly lower than the expected doubling of the SAR (1.3% overall, p<0.0001; and 2.9% for CR, p = 0.035). Comparisons of observed and expected SAR using the proof-of-concept study outcome definitions were also statistically significant (p<0.0001, S6 Text).
Discussion
In this section, the researchers explain that they tried as best as possible to emulate environmental conditions that would be seen during the winter months when infections are at their peak. They even attempted to maintain low humidity in order to increase transmission. However, the experiment resulted in the “near absence” of transmission. They had to conclude that contact and large droplet spray did not contribute to infection in these optimal settings. In fact, they admitted that their findings did not allow for any definitive conclusion as to how influenza supposedly spreads.
Even though they doubled the duration of the exposure and the amount of donors, the researchers could not meet the secondary attack rate that they had expected to see. They attempted to blame this on the outcome criteria, as in the proof-of-concept study, only a single NPS positive by qRT-PCR without seroconversion was required. In the present study, they required two or more NPS positive by qRT-PCR in the absence of seroconversion. As they could not achieve transmission with their selected conditions, the researchers thought of various ways that they could try and create more infections in the future, including the type of “virus” used, the route of inoculation, susceptibility of the human volunteers, the rate of “viral” shedding into NPS and aerosols, and reducing ventilation of exposure rooms.
To our knowledge, this is the largest human influenza challenge-transmission study undertaken to date. We applied measures to control and standardise environmental conditions and ventilation rates within and between exposure events, to emulate as far as possible indoor winter conditions when respiratory virus spread is maximal. We particularly sought to maintain low humidity conditions which have been associated with enhanced transmission [13] and increased virus viability [14], together with a low ventilation rate to maximize recipient exposure to airborne virus. The near absence of transmission to control Recipients suggests contact and large droplet spray did not contribute substantially to transmission under the conditions used in these EEs. The significantly lower than expected SAR in this study compared with the proof-of-concept study, which had much lower ventilation rates, suggests aerosols as an important mode of influenza virus transmission in this model. The overall low SAR suggests that Donors in this model were minimally contagious and prevents definitive assessment of the modes of transmission.
Having reported an SAR of 25% (3/12) in our earlier proof-of-concept study, we expected to observe an SAR of >25%, having doubled both the duration of the exposure and the number of Donors in each quarantine [15]. Indeed, the study was designed to examine an SAR of 40% in CR versus 20% in IR which would have required 125 Recipient volunteers; this was not met (n = 75) and the study was underpowered.
However, the outcome criteria used in the proof-of-concept study, which included as positive a single NPS positive by qRT-PCR without seroconversion, were made more stringent in the present study by requiring two or more NPS positive by qRT-PCR in the absence of seroconversion (S6 Text). Applying the proof-of-concept criteria to the current study gives an SAR of 4% (3/75) overall, while applying the stricter criteria used in this study to the proof-of-concept study gives an SAR of 8.3% (1/12) rather than 25%. Given the lower than planned enrolment and the stricter outcome criteria, this study was doubly underpowered.
These observations raise two questions: 1) Why were SARs, using stringent criteria, low in both studies and what are the implications for future human challenge-transmission studies? 2) Why was the SAR significantly lower in the present study compared with the expected doubling of the rate observed for the proof-of-concept?
The low SAR in these studies suggests that, unless a much greater SAR can be achieved, type II error associated with underpowering will be a major obstacle to successful use of human challenge-transmission studies. Potential areas to consider addressing in order to raise the SARs in future studies include the virus used, the route of inoculation, susceptibility of the human volunteers, the rate of viral shedding into NPS and aerosols, and reducing ventilation of exposure rooms.
As far as the “virus” utilized in their challenge study, it was stated that they used a GMP A/WI challenge “virus” manufactured by Baxter BioScience (Vienna, Austria). Stocks of this “virus” preparation were evaluated in a separate 2016 study that was linked. From this 2016 reference, we can see that they did not take a sample directly from a sick human and use purified and isolated “viral” particles in an attempt to infect other humans naturally. What they used instead was a creation from an original sample that was passaged 3 times in avian primary chicken kidney (CK) cells, 4 times in embryonated chicken eggs, and then twice in GMP Vero cells to generate the “viral” stock that was used to inoculate intranasally the challenge study subjects. Sequencing data told them that it “was at least partially adapted” to the egg and/or tissue environments in which it was produced. In other words, it contained chicken and monkey kidney contaminants. What exact chemicals and media used were not listed. As usual, the “virus” is nothing but a toxic cell culture soup that was administered in an unnatural way through the nose.
Deep Sequencing of Influenza A Virus from a Human Challenge Study Reveals a Selective Bottleneck and Only Limited Intrahost Genetic Diversification
“Production of challenge virus.
The virus used for experimental challenge was produced under current good manufacturing practices (cGMP) at Baxter Bioscience (Vienna, Austria). The original virus was obtained from a human isolate of A/Wisconsin/67/2005 (H3N2) (GenBank accession numbers CY114381 to CY114388). We here refer to this human isolate as the “reference strain.” This virus was passaged 3 times in avian primary chicken kidney (CK) cells, 4 times in embryonated chicken eggs, and then twice in GMP Vero cells to generate the viral stock used to inoculate the challenge study subjects (Fig. 1).”
https://journals.asm.org/doi/10.1128/JVI.01657-16
The researchers admitted that they only observed symptoms ranging from asymptomatic (i.e. none) to febrile symptomatic infection, which usually consists of a fever. However, no fevers were observed in this study. They attempted to blame “positive selection” of the challenge “virus” for growth in the production environment, rather than for human transmissibility, as a reason for why their study was unsuccessful. They also blamed the route of infection, stating that experimental infection with aerosolized “virus” at lower doses was more likely to result in ‘typical influenza-like disease’ (fever plus cough) than was intranasal inoculation. Even though the exact correlates of immunity and severity using novel immunological assays had not been validated, the researchers could not blame their antibody results since prior immunity, as measured by the HAI and MN assays, ruled it out. Thus, they concluded that antibodies were not a major limitation and could not account for their failure to transmit from readily infected individuals to identically screened uninfected individuals.
In the proof-of-concept (2009) and follow-on (2013) studies we used a GMP A/WI challenge virus manufactured by Baxter BioScience (Vienna, Austria). Both studies produced similar clinical and serological infection rates (typically 60–70% and 70–75%, respectively) after inoculation via nasal instillation, and similar spectrums of clinical illness severity in Donors. These rates were higher than reported by a previous study using lower doses of the same viral preparation [16] and consistent with rates reported in a review of 56 challenge studies [17]. The illnesses we observed were similar to the range seen in healthy adults in the community, from asymptomatic to febrile symptomatic infection [18]. Thus, skewed illness severity does not seem to explain the low SAR.
The virus preparation has been used in other human challenge studies with similar rates of infection via nasal instillation [19]. Using deep sequencing, Sobel Leonard and colleagues showed that a sample of the Baxter stock “was at least partially adapted” to the egg and/or tissue environments in which it was produced [19]. They also found that nasal instillation of the stock into human volunteers resulted in rapid purification selection, although a fixed variant in the HA gene remained. We have performed a BLAST search and identified the fixed variant (G70A/D8N) in deposited sequences of wild-type H3N2 viruses. This suggests that, on its own, the fixed HA variant is unlikely to have been a key alteration. These results suggest that it is unlikely that the virus stock was the primary cause of the low SARs. But, the impact of positive selection of the challenge virus for growth in the production environment, rather than for human transmissibility, remains a potential contributing factor to consider in choosing challenge viruses for future transmission studies.
The route of infection with influenza virus is known to matter in the setting of experimental infection, with aerosolized virus infectious at lower doses and more likely to result in ‘typical influenza-like disease’ (fever plus cough) than intranasal inoculation [20,21]. This anisotropic property [22] of influenza virus is not unique among respiratory viruses; e.g. it is exploited by the live, unattenuated adenovirus vaccine [23]. The implication for human challenge-transmission studies, however, may be that increased rates of lower respiratory tract infection via aerosol inoculation might be required to achieve sufficiently high rates of donors with fever, cough, and contagiousness to achieve a useful SAR.
In the current quarantine-based human challenge-transmission model, consistent with historical precedent, screening for susceptibility was undertaken primarily by HAI antibody screening, although it is recognised that screening by MN titre or other assays [24,25] could be an alternative or adjunctive approach. The exact correlates of immunity and severity using novel immunological assays have not been validated and selecting subjects based on these assays would have added substantial complexity and costs. Six Donors and five Recipients in the present study were discovered, in retrospect, to have seroconverted during the 3 to 56-day interval prior to entering the quarantine facility, despite having as short a delay as possible between final screening for HAI and quarantine entry. However, the majority of Donors and Recipients were susceptible according to the results of microneutralization tests. Prior immunity, as measured by the HAI and MN assays, does not therefore, appear to have been a major limitation nor account for failure to transmit from readily infected Donors to identically screened Recipients. Regarding future studies, as novel immune correlates of influenza protection and severity become established, additional approaches beyond HAI and MN assays could be employed for volunteer selection. This might enable selection of those likely to become infected, febrile, and have greater symptomatology including more frequent and greater levels of cough and runny nose. Unfortunately, such screening might also dramatically reduce the yield of suitable volunteers and substantially increase overall study costs.
Interestingly, the researchers measured “viral” load via qRT-PCR and noted that the amount of “virus” present was sufficiently high. Thus, it could not be stated that low “viral” secretions from nasal discharge could be blamed for the failure to transmit infection. It was also noted that 26% of the “infected” donors had detectable “virus” in their breath samples. When compared with “naturally infected” influenza cases, the experimentally “infected” individuals shed less “virus” than their counterparts. Thus, the researchers hypothesized that aerosols largely derived from the lower respiratory tract will lead to an increase in the frequency of lower respiratory tract infection and that future challenge studies needed to reflect this guess. The researchers also decided that their failure may have been due to different ventilation conditions from their proof-of-concept study that may not have allowed for successful aerosol infection in the current study. They felt that this possibility would be especially important if the “infected” in future studies continued to represent the lower end of the aerosol shedding spectrum not observed in “naturally infected” cases. While ventilation was considered a possible reason for their failure, the humidity in the hotel rooms was not. The researchers attempted to achieve conditions that led to volunteer comfort as well as those favorable to “transmission.”
Although their study was a failure, the researchers tried their best to point out “important findings” from this experiment. They were as follows:
- Even though fewer “viral” challenged subjects had “virus-laden” aerosols than seen in people with “natural infections,”those volunteers who did produce “viral” aerosols did so at a rate similar to the average symptomatic “naturally infected” case
- Observation of transmission via aerosols in quarantine studies may be strongly dependent on the dilution ventilation rate
- Low risk of transmission to control recipients suggested that contact and large droplet spray transmission were not important modes of transmission in this model
- Aerosol transmission may be an important mode of influenza “virus” transmission between adults
- However, sensitivity to experimental conditions demonstrated that it will be challenging to generalize the results of the quarantine-based transmission model to broad conclusions about the relative importance of aerosol, droplet spray, and contact modes of transmission
- Although an important role for aerosols in transmission of influenza is hinted at when comparing the proof-of-concept and current studies, this challenge model cannot provide a definitive answer to the importance of this mode for influenza “virus” transmission between humans
Results from serial nasopharyngeal swabs in Donors indicate that over 80% were positive by qRT-PCR testing on one or more post-challenge days. Viral load detected by swabs was substantial with qRT-PCR Ct values in the mid 20s on days two and three (Fig 3). Thus, failure to shed virus into nasal secretions cannot explain the low SARs. Minimal transmission despite days of prolonged exposure to Donors shedding virus into nasal secretions provides strong evidence that contact and large droplet transmission are not important in this model.
The results from breath sampling with the Gesundheit-II device indicate that 26% (11/42) of infected Donors had virus detectable in exhaled air during the same period. By comparison, virus shedding into exhaled breath was detected in 84% (119 of 142) of influenza cases selected on the basis of having fever or a positive rapid test and sampled on one to three days post onset of symptoms, mostly recruited from young adults on a college campus [7]. When compared on a per-sample basis, infected Donors shed detectable virus less frequently than naturally infected college campus cases [7] in both coarse (7% and 40%, respectively) and fine aerosols (15% and 76%); all assays for both groups were performed in the same laboratory using the same methods. However, when the comparison was limited to positive aerosol samples from each study population, the average quantities of virus detected were similar (within 1 log), for the Donors as for the college community cases (GM coarse 3.1E+3 and 1.2E+4, GM fine 5.3E+3 and 3.8E+4, respectively). The maximum exhaled breath viral aerosol from the 11 Donors was two to four logs lower than from the college campus cases selected for having fever or a high viral load in the NPS (maximum coarse 2.8E+4 and 4.3E+8, and maximum fine 8.0E+4 and 4.4E+7, respectively) [7]. While this difference may merely represent the low probability of sampling from the tail of a log-normal distribution with only 11, as compared with 119 cases, it may be relevant to the low SAR in the challenge-transmission model if aerosols disseminated by more symptomatic individuals, such as the selected community cases, and rare supershedders account for most transmission. If aerosols are largely derived from the lower respiratory tract, as has been suggested by analysis of the college community cases, this would also suggest that future challenge-transmission studies should employ methods designed to increase the frequency of lower respiratory tract infection.
The proof-of-concept study was conducted in a hotel room with closed windows and thermal control provided only by a recirculating air conditioning unit. While the ventilation rate was not measured, it was likely to have been extremely low for the number of occupants, with only small, intermittent, bathroom extract and natural building infiltration providing fresh air. The ventilation rate of 4 l/s/person during the main study was low compared to 10 l/s/person recommended in UK design standards [26] but was likely substantially greater than in the proof-of-concept study. This would have produced significantly higher viral aerosol concentrations during the proof-of-concept EEs, assuming similar generation rates from Donors in both experiments. Given that the Donors in the two studies were similar in other respects, differences in shedding rates seem unlikely. Therefore, the difference in SAR between this study and the expected SAR based on design changes and prior results are possibly due to differing ventilation conditions. The implication for future challenge-transmission studies, given that the ventilation rate in the current study was as low as possible with a single pass ventilation system, is that recirculating air conditioning systems similar to that in the proof-of-concept study should be employed to limit dilution ventilation and maximize exposure to aerosols. This will be especially important if Donors in future studies continue to represent the lower end of the aerosol shedding spectrum seen in naturally infected cases.
Achieving temperature and humidity to simulate winter conditions was challenging, particularly in Q3, conducted in June 2013 when the average external conditions were 16°C and 64% relative humidity. It was necessary to strike a balance between volunteer comfort and conditions favourable to transmission, both of which were constrained by the capability of the mechanical systems in the building. However, the relative humidity in the current study overlapped with that during the proof-of-concept study, which ranged between 38 and 53%, and thus, eliminated humidity as a potential explanation for the difference in transmission rates.
Despite this study not having produced the planned SAR, it yields important findings. First, although fewer viral challenged subjects had virus-laden aerosols than seen in people with natural infections presenting with influenza-like symptoms, those volunteers who did produce viral aerosols did so at a rate similar to the average symptomatic naturally infected case. Second, given that a subset of the infected volunteers had moderate viral aerosol shedding in this model, observation of transmission via aerosols in quarantine studies may be strongly dependent on the dilution ventilation rate. Third, low risk of transmission to Control Recipients suggests that contact and large droplet spray transmission were not important modes of transmission in this model. The overall low SAR compared to that observed in the proof-of-concept study suggests that, given the main difference between the studies was the indoor air ventilation rate, aerosol transmission may be an important mode of influenza virus transmission between adults. Finally, sensitivity of transmission to details of the Donors selected, environment, and activity during exposure events, suggest that if a successful transmission model can be developed, carefully designed studies may be useful for investigating specific, targeted intervention strategies for prevention of specific transmission modalities. However, sensitivity to experimental conditions also demonstrates that it will be challenging to generalize the results of the quarantine-based transmission model to broad conclusions about the relative importance of aerosol, droplet spray, and contact modes of transmission. These complexities of the challenge-transmission model suggest that community-based transmission studies employing deep-sequencing based molecular epidemiologic methods in natural experiments, e.g. comparing high and low ventilation dormitories or barracks, may be more attractive alternatives than previously thought. Unfortunately, although an important role for aerosols in transmission of influenza, at least between adults, is hinted at when comparing the proof-of-concept and current studies, this challenge model cannot provide a definitive answer to the importance of this mode for influenza virus transmission between humans.
Materials and Methods
I’m only hightling a few areas from the methods section to emphasize some points. First, the intervention recipients (IR’s) wore face shields, hand sanitized once every 15 minutes, and could only touch their faces with once-use wooden spatulas. This was to ensure that they could only come into contact with aerosolized “virus.” The control recipients could apparently do whatever they wanted.
Second, as stated earlier, the “virus” was a lab-created concoction manufactured and processed under current good manufacturing practices (cGMP). This goo was inoculated intranasally, which is not a natural route of exposure and infection.
Third, individual donors and recipients were each set up in a single exposure room per day where they interacted at close distances for approximately 15 hours a day for four consecutive days. This should make one question how “contagious” this lab-created goo truly is if only one person was potentially “infected” via airborne particles.
Fourth, the rooms were adjusted in order to create conditions favorable to influenza transmission. The temperature, humidity, and ventilation were all carefully controlled in order to increase transmission, and yet this did not occur, and the study was considered a failure.
Fifth, all participants had to undergo monitoring of respiratory and systemic symptoms three times a day. Nasopharyngeal swabs were done daily. Despite this intense monitoring, no transmission occurred.
Finally, influenza-like Illness (ILI) was defined as an illness >24 hours duration with: either fever and at least one respiratory symptom. That seems like a really loose definition of influenza, and yet, the study failed to achieve its intended goals even with the watered-down criteria.
Overview
Volunteers, screened for serologic susceptibility, were randomly selected for intranasal challenge with A/WI [15]–becoming ‘Donors’. After allowing for a short incubation period, Donors were introduced to other sero-susceptible volunteers–‘Recipients’–under controlled household-like conditions for four days. Recipients were randomised as Intervention Recipients (IR) or Control Recipients (CR). IRs wore face shields evaluated to interrupt large droplet transmission but to be permissive to aerosols (S3 Text); in addition, IRs hand sanitised (using alcohol-based Deb InstantFOAM, 72% ethyl alcohol) once every 15 minutes to minimise the possibility of contact transmission. IRs were only allowed to touch face via single-use wooden spatulas.Thus, IRs would be exposed to influenza only via aerosols. CRs did not wear face shields or use hand sanitiser and were allowed to touch face freely; therefore, CRs would have been exposed via all routes of transmission consistent with close proximity human-human contact. Fig 1 shows an overview of the study design.
Influenza virus
Influenza A/WI manufactured and processed under current good manufacturing practices (cGMP) was obtained from Baxter BioScience, (Vienna, Austria). Stocks of this virus preparation have been sequenced and its evolution in the upper respiratory track of inoculated volunteers extensively analysed [19].”
Exposure events
“The study was conducted in three, separate, identically-designed quarantine events (Q1, Q2, and Q3). From Day 1 to Day 4 of each quarantine event, all volunteers took part in an Exposure Event (EE). Individual Donors and Recipients were each allocated to a single exposure room per day where they interacted at close distances for approximately 15 hours/day, for four consecutive days. In-room staff supervised activities such as playing board games, pool, and table football, and watching films, whilst ensuring that volunteers mixed freely, and that IRs complied with face shield use, hand sanitisation, and no-touch-face rules. Donor, IR and CR groups were moved into different corners of the rooms for meal breaks, and Donor and Recipient groups were housed separately at night, including further separation and withdrawal of any Recipients with symptoms to prevent any contamination of the results by Recipient-Recipient secondary transmission. Five exposure rooms were used ranging from 17-30m2 floor area and 50-87m3 volume. Four Donors were non-randomly allocated to each exposure group to ensure even distribution of subjects actively shedding virus. This was achieved by assessing symptom scores and the results of influenza rapid tests (Quidel Sofia) performed on Days 1 and 2. From Day 2 onwards, Donors remained in their allocated group and were not redistributed further. Once assigned to an exposure group, Recipients remained in the same group until the end of the EE or until they developed ILI and were withdrawn to a separate isolation area. On each day of EE, each exposure group rotated to a different exposure room.
Environmental controls
Each exposure room was assessed pre-quarantine by building and ventilation engineers and modified to achieve a ventilation rate of approximately 4L/second/person (based on planned occupancy during EEs), temperature 18–22°C, and relative humidity 45–65%, to produce conditions favourable to influenza transmission [13], balanced against tolerability for occupants, and the capability of building systems to provide controlled environments comparable across all three quarantine studies. During each EE, rooms were monitored at 5-minute intervals for CO2 concentration (as a proxy for ventilation rate), temperature and humidity; heating, cooling and humidity were remotely adjusted to maintain optimal conditions.
Clinical assessment
All subjects underwent thrice daily monitoring of respiratory and systemic symptoms; each symptom was reported as grade 0 (not present) to 3 (severe). Paired venous blood specimens for serology were taken on Day -2 and Day 28. NPS were taken daily from all subjects. Respiratory specimens were analysed by quantitative reverse-transcriptase PCR (qRT-PCR) and serological specimens by HAI and microneutralisation (MN) assays. The qRT-PCR and HAI were performed in duplicate at the MRC University of Glasgow Centre for Virus Research (with Fast Track Diagnostics qRT-PCR kit) and the U.S. Centers for Disease Control and Prevention (CDC), Atlanta; MN assays were performed by CDC as described previously [28].”
Outcome definitions
“Respiratory symptoms were defined as self-reported grade ≥1 of runny nose, stuffy nose, sneeze, sore throat, cough, or shortness of breath ‘lasting ≥24 hours’ (S4 Text). Fever was defined as temperature >37.9 oC. Symptomatic was defined as evidence of any respiratory symptom lasting ≥24 hours during study days 1–6. Influenza-like Illness (ILI) was defined as an illness >24 hours duration with: either fever and at least one respiratory symptom; or two or more symptoms of grade ≥1, one of which must have been respiratory; eligible non-respiratory symptoms were headache, muscle/joint ache, and malaise; lasting ≥24 hours means report of a respiratory symptom during 3/3 observations within a single day, or at least once per day over two consecutive days. Laboratory confirmed infection was defined as: a 4-fold or greater rise in HAI or MN titres between Day -2 (baseline) and Day 28; or two or more positive NPS test results by qRT-PCR. These differed from the proof-of-concept study, which used seroconversion or a single positive nasal wash (S6 Text).”
https://journals.plos.org/plospathogens/article?id=10.1371/journal.ppat.1008704
In Summary:
- Uncertainty about the importance of influenza transmission by airborne droplet nuclei generates controversy for infection control
- Only one “transmitted infection” was confirmed by antibody testing in a control, yielding a secondary attack rate of 2.9% among the control and 1.3% overall, significantly less than 16% (p<0.001) expected based on a proof-of-concept study secondary attack rate and considering that there were twice as many donors and days of exposure
- The authors admit that human-to-human transmission remains poorly understood
- The relative importance of the different modes of transmission (direct and indirect contact, large droplet, and aerosols (airborne droplet nuclei)) remains uncertain during symptomatic and asymptomatic infection
- The current evidence for transmission comes from indirect means such as:
- “Virus deposition and survival in the environment
- The epidemiology of disease
- Pharmaceutical and non-pharmaceutical interventions
- Animal models
- Mathematical models of transmission
- Infection control guidance for pandemic and seasonal influenza assumes that most transmission occurs during symptomatic infection, predominantly via large droplet spread at short range (i.e. they have no clue nor any scientific evidence supporting their assumption)
- An expert panel, after in-depth review of the challenges facing community and workplace-based intervention studies and their failure thus far to provide definitive evidence regarding the relative contribution of the various modes of transmission, concluded that a human challenge-transmission study would be a more promising direction for future research
- Challenge studies assessing human-to-human transmission had not been performed (I guess they would like to conveniently ignore Rosenau’s studies and those that came afterward in the early 1900’s 🤷♂️)
- A proof-of-concept study in 2009 suggested that larger scale human challenge-transmission models might be useful to evaluate transmission modes
- This was the large-scale version of the 2009 study, and it did not achieve the intended level of transmission required for a more conclusive interpretation
- No serious adverse events were recorded in any of the volunteers who commenced the study
- The unnatural method of intranasal inoculation produced an “infection” rate of 81% (42/52) among inoculated volunteers
- Of the 42 “lab-confirmed” infected donors 25 (60%) had ILI and 10 (24%) were classified as asymptomatic
- Only 36 of 52 donors had nasopharyngeal swab (NPS) that tested positive by PCR
- They “observed” aerosol shedding from only 11 (26%) of the 42 successfully “infected” donors
- None of the recipients developed fever
- Only one secondary “infection” was confirmed by antibody testing
- One control subject was symptomatic, and the symptoms met the definition of ILI, but the qRT-PCR evaluations were persistently negative
- Two other control patients were transiently qRT-PCR positive, but neither met laboratory positivity criteria
- The observed secondary attack rate (SAR) for the current study was not significantly different than that of the adjusted SAR from the proof-of-concept study but was significantly lower than the expected doubling of the SAR
- The researchers applied measures to control and standardise environmental conditions and ventilation rates within and between exposure events, to emulate as far as possible indoor winter conditions when respiratory “virus” spread is maximal
- The near absence of transmission to control recipientssuggested contact, and large droplet spray did not contribute substantially to transmission under the conditions used in these EEs
- The overall low SAR suggested that donors in this model were minimally contagious and prevented definitive assessment of the modes of transmission
- They expected to observe an SAR of >25% as they doubled both the duration of the exposure and the number of donors in each quarantine yet did not come close to achieving this outcome
- This led the researchers to 2 questions:
- Why were SARs, using stringent criteria, low in both studies and what are the implications for future human challenge-transmission studies?
- Why was the SAR significantly lower in the present study compared with the expected doubling of the rate observed for the proof-of-concept?
- The researchers used a “Good Manufacturing Practice” (GMP) A/WI challenge “virus” manufactured by Baxter BioScience (Vienna, Austria)
- From a linked 2016 study, we find that the “virus” was a creation stemming from being passaged 3 times in avian primary chicken kidney (CK) cells, 4 times in embryonated chicken eggs, and then twice in GMP Vero cells to generate the “viral” stock used to inoculate the challenge study subjects
- The “virus” was “at least partially adapted” to the egg and/or tissue environments in which it was produced (i.e. it contained chicken and monkey contaminants)
- The illnesses observed were similar to the range seen in healthy adults in the community, from asymptomatic to febrile symptomatic infection
- The researchers attempted to blame “positive selection” of the challenge “virus” for growth in the production environment, rather than for human transmissibility, for their failure
- They also attempted to blame the route of infection with influenza “virus” with aerosolized “virus” infectious at lower doses being more likely to result in ‘typical influenza-like disease’ (fever plus cough) than intranasal inoculation
- It was admitted that the exact correlates of immunity and severity using novel immunological assays have not been validated
- Prior immunity, as measured by the HAI and MN assays, was not a major limitation, nor could it account for their failure to transmit from readily infected donors to identically screened recipients
- They felt that, as novel immune correlates of influenza protection and severity become established, they might be better able to screen individuals
- “Viral” load detected by swabs was substantial with qRT-PCR Ct values in the mid 20s on days two and three
- Thus, failure to shed “virus” into nasal secretions cannot explain the low SARs
- The results from breath sampling with the Gesundheit-II device indicated that 26% (11/42) of infected donors had “virus” detectable in exhaled air during the same period
- They concluded that ventilation in the hotel rooms would have produced significantly higher “viral” aerosol concentrations during the proof-of-concept EEs, assuming similar generation rates from donors in both experiments
- They decided that the difference in SAR between this study and the expected SAR based on design changes and prior results were possibly due to differing ventilation conditions
- The researchers felt that ventilation would be important to future studies if the experimentally “infected” continued to represent the lower end of the aerosol shedding spectrumin comparison to “naturally infected” cases
- As it was necessary to strike a balance between volunteer comfort and conditions favourable to transmission, they eliminated humidity as a potential explanation for the difference in transmission rates
- Despite their failure, the researchers noted “important findings” from their research:
- Although fewer “viral” challenged subjects had “virus-laden” aerosols than seen in people with “natural infections” presenting with influenza-like symptoms, those volunteers who did produce “viral” aerosols did so at a rate similar to the average symptomatic naturally infected case
- Observation of transmission via aerosols in quarantine studies may be strongly dependent on the dilution ventilation rate
- Low risk of transmission to control recipientssuggested that contact and large droplet spray transmission were not important modes of transmission in this model (i.e. they could not spread the “virus” through the two main modes of transmission)
- The overall low SAR compared to that observed in the proof-of-concept study suggests that, given the main difference between the studies was the indoor air ventilation rate, aerosol transmission may be an important mode of influenza “virus” transmission between adults
- Sensitivity to experimental conditions demonstrated that it will be challenging to generalize the results of the quarantine-based transmission model to broad conclusions about the relative importance of aerosol, droplet spray, and contact modes of transmission
- Although an important role for aerosols in transmission of influenza, at least between adults, is hinted at when comparing the proof-of-concept and current studies, this challenge model could not provide a definitive answer to the importance of this mode for influenza “virus” transmission between humans
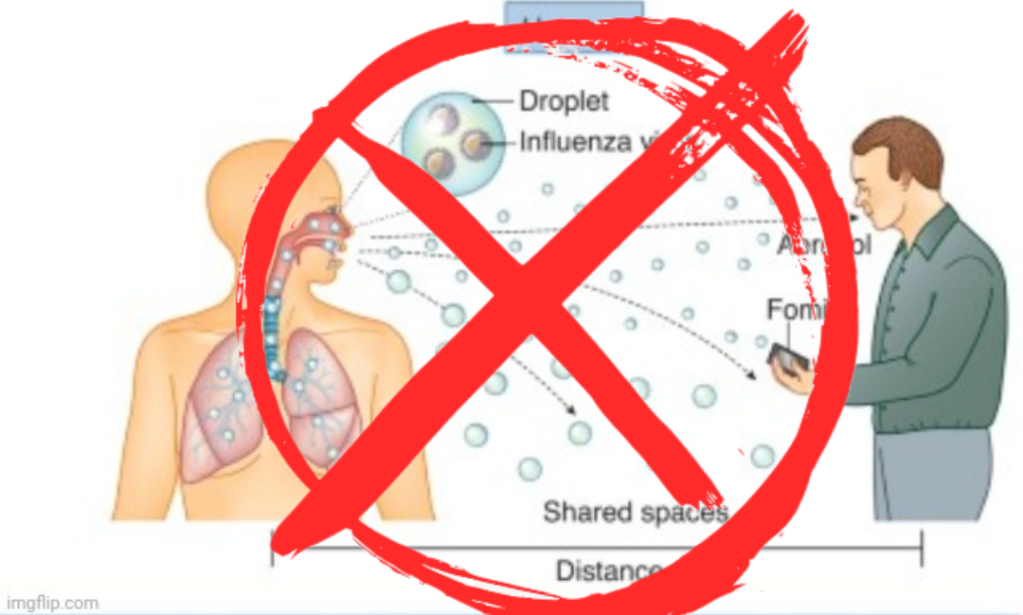
We are now over a century into investigating the influenza “virus,” and researchers are still unable to definitively say how the “virus” spreads. Virologists have repeatedly failed in the past to prove human-to-human transmission via “natural” routes of exposure, and even under highly controlled conditions using lab-created “viral” soup injected intranasally into volunteers, only 25 of 52 had “influenza-like” symptoms. These symptoms consisted mainly of runny nose, stuffy nose, sneeze, sore throat, cough, or shortness of breath lasting ≥24 hours. Logically, these symptoms would be the expected reactions to having chemical goo injected into the nose that seeps down the throat and into the respiratory tract. None of those who were “infected” via intranasal injection developed a fever, and 10 of the 42 lab-confirmed cases were asymptomatic. Only one secondary “infection” was confirmed by antibody testing as the gold standard PCR test failed to detect it. In fact, only 36 of 52 of the donors had nasopharyngeal swabs (NPS) that tested positive by PCR. In other words, while the researchers attempted to claim successful “infection” via an unnatural route of “infection” with evidence based upon indirect means such as PCR results, antibody measurements, and vague symptoms (or even a lack of symptoms), they could not definitively show human-to-human transmission. The researchers could only show one secondary “infection,” which was statistically insignificant and could have easily been due to errors in the admittedly unreliable antibody measurements. This failure to show secondary transmission happened in highly controlled conditions meant to enhance this supposed transmission, which makes this inability to transmit “infection” even more damning for virology.
Thus, even though we are well over a century into investigating influenza, there is still no scientific evidence of human-to-human transmission. In fact, there is plenty of evidence that this does not occur. With this abject failure to prove contagion, we are left with pseudoscientific evidence such as “virus” deposition and survival in the environment; the epidemiology of disease; pharmaceutical and non-pharmaceutical interventions; animal models; and mathematical models of transmission as the means to create this fictional narrative to convince the uninformed that such a process occurs. In light of this failure to prove human-to-human transmission of influenza in humans, it is safe to conclude that Bridges et al. 2003 statement still stands:
There are no human experimental studies published in the English-language literature delineating person-to-person transmission of influenza.
Source: https://viroliegy.com/2023/04/16/challenging-influenza/